(Redirected from Niobium(V) iodide)
Jump to navigationJump to search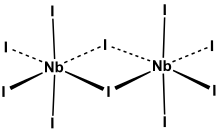 | |
| Names | |
|---|---|
| Other names Niobium(V) iodide | |
| Identifiers | |
3D model (JSmol) | |
| ChemSpider | |
| ECHA InfoCard | 100.034.006 |
| EC Number |
|
PubChem CID | |
CompTox Dashboard (EPA) | |
| |
| |
| Properties | |
| Nb2I10 | |
| Molar mass | 1475 |
| Appearance | yellow solid |
| Density | 5.30 g/cm3 |
| Melting point | 543 °C (1,009 °F; 816 K) sublimes |
| Hazards | |
| GHS pictograms |  |
| GHS Signal word | Danger |
| H314, H318 | |
| P260, P264, P280, P301+330+331, P303+361+353, P304+340, P305+351+338, P310, P321, P363, P405, P501 | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Niobium(V) iodide is the inorganic compound with the formula Nb2I10. Its name comes from the compound's empirical formula, NbI5.[1] It is a diamagnetic, yellow solid that hydrolyses readily. The compound adopts an edge-shared bioctahedral structure, which means that two NbI5 units are joined by a pair of iodide bridges. There is no bond between the Nb centres.[2] Niobium(V) chloride, niobium(V) bromide, tantalum(V) chloride, tantalum(V) bromide, and tantalum(V) iodide, all share this structural motif.
Synthesis and structure[edit]
Niobium pentaiodide forms from the reaction of niobium with iodine:
- 2 Nb + 5 I2 → 2 NbI5
The method used for the preparation of tantalum(V) iodide using aluminium triiodide fails to produce pure pentaiodide.[3]
References[edit]
- ^ Greenwood, N. N.; & Earnshaw, A. (1997). Chemistry of the Elements (2nd Edn.), Oxford: Butterworth-Heinemann. ISBN 0-7506-3365-4.
- ^ Krebs, Bernt; Sinram, Diethard "Darstellung, Struktur und Eigenschaften einer neuen Modifikation von NbI5 (Preparation, structure and properties of a new modification of NbI5" Zeitschrift fǔr Naturforschung, Teil B: Anorganische Chemie, Organische Chemie 1980, volume 35b, pp. 12-16.
- ^ G. Braurer (1963). "Niobium(V) Iodide". In G. Brauer (ed.). Handbook of Preparative Inorganic Chemistry, 2nd Ed. 1. NY, NY: Academic Press. p. 1315.